Therapeutic H2S Delivery:
Hydrogen sulfide (H2S) is known primarily as a foul-smelling, toxic pollutant. However, H2S is also a vital biological signaling gas, produced by enzymes in our bodies. In fact, along with carbon monoxide (CO) and nitric oxide (NO), H2S is recognized as the third member of the class of signaling gases known as gasotransmitters. The physiological roles of H2S are still being discovered, and it is of interest as a potential therapeutic in a wide variety of diseases and conditions. However, the majority of biological studies on H2S have been carried out with systemically administered small molecule H2S donors, primarily Na2S and NaHS, which have little tissue specificity and the potential for off-target effects. As a result, delivering H2S to a desired site of action at therapeutic dosages remains difficult, and new methods to control the dosage, rate, location, and timing of H2S delivery are needed (Figure 1).
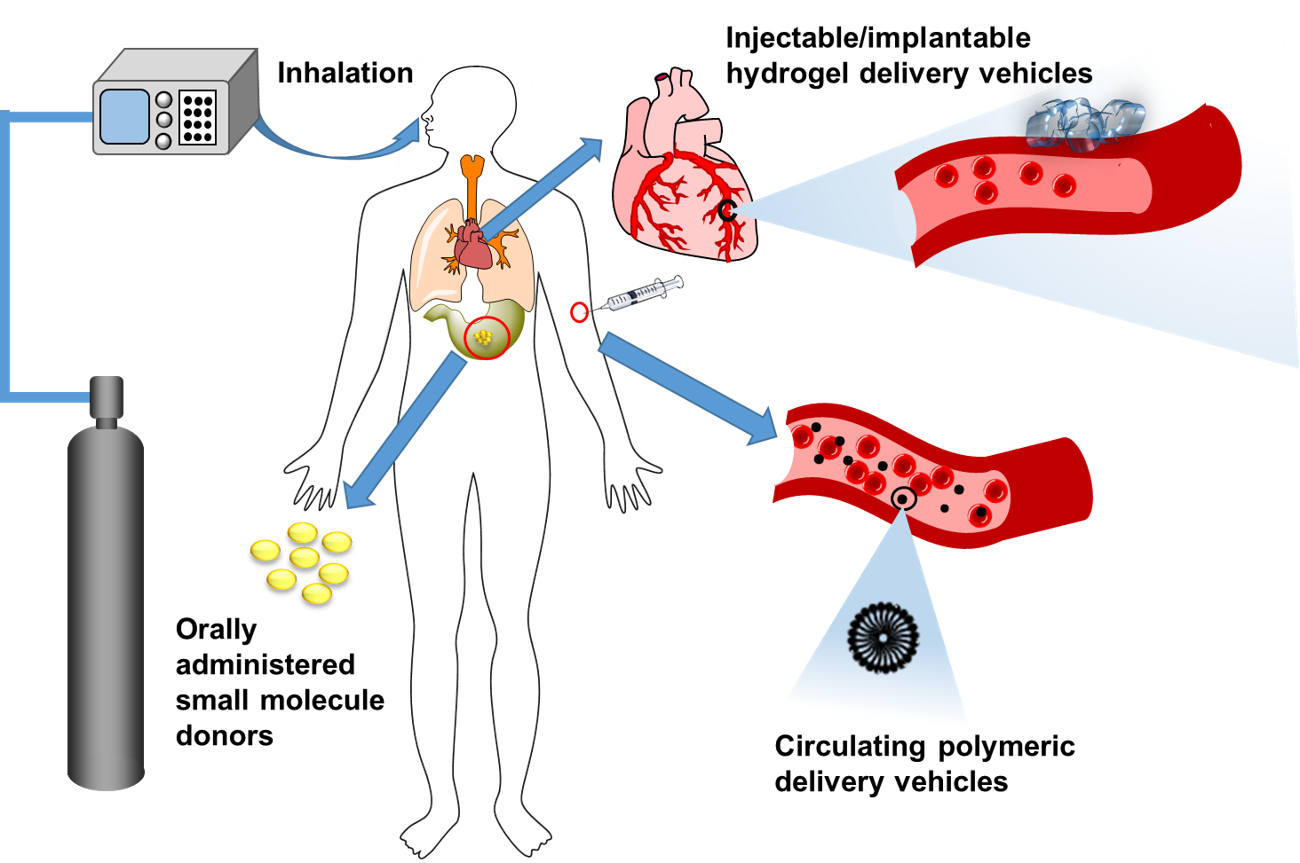
Figure 1. Methods of H2S delivery. Reproduced from Qian, Y.; Matson, J. B. “Gasotransmitter delivery via self-assembling peptides: Treating diseases with natural signaling gases” Adv. Drug Deliv. Rev. 2017, 137, 110-111.
We focus on the development of small molecules, polymers, gels, and polymer assemblies that release H2S and related reactive sulfur species (RSS) in response to specific triggers. These compounds and materials allow us to control the delivery of H2S/RSS in order to study their (patho)physiological roles and evaluate their therapeutic potential.
Click here to see a recent video on our H2S work.
Bottlebrush Polymers:
The expansion of controlled polymerization techniques over the past two decades has enabled the construction of polymers with complex topologies. Bottlebrush polymers contain a polymer backbone with densely grafted polymer side chains, which force the backbone polymer into an extended conformation. These types of polymers may have applications as supersoft elastomers, vibration damping materials, and as nano-objects of controlled dimensions. We are pursuing new synthetic routes to bottlebrush polymers using a variety of polymerization techniques, including reversible addition-fragmentation transfer (RAFT) polymerization, atom transfer radical polymerization (ATRP), and ring-opening metathesis polymerization (ROMP).
In particular, we are interested in preparing bottlebrush polymers with shape asymmetry. Our current interest is in polymers that are cone-shaped, which may have interesting rheological and self-assembly properties. We synthesize these cone-shaped polymers, termed tapered bottlebrush polymers, via a grafting-through strategy termed sequential addition of macromonomers ring-opening metathesis polymerization (SAM-ROMP). Using this method, we have prepared bottlebrush polymers with systematically varied side-chain molecular weights ranging from 1–10 kg/mol (Figure 2).
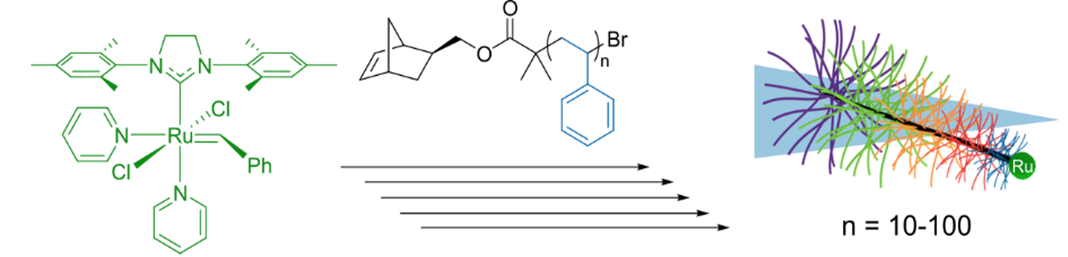
Figure 2. Synthesis of tapered bottlebrush polymers via SAM-ROMP. Reproduced from Radzinski, S. C.; Foster, J. C.; Scannelli, S. J.; Weaver, J. R.; Arrington, K. J.; Matson, J. B. “Tapered Bottlebrush Polymers: Cone-Shaped Nanostructures by Sequential Addition of Macromonomers” ACS Macro Lett. 2017, 6, 1175-1179.
We are also interested in understanding how bottlebrush block copolymers self-assemble in solution, and how their assemblies vary compared with traditional linear block copolymers. In this project area, we are preparing bottlebrush block copolymers from hydrophobic and hydrophilic macromonomers using ROMP techniques. We allow the micelles to self-assemble in water and then characterize them using a variety of techniques, including microscopy, light scattering techniques, surface tension measurements, and small-angle neutron scattering (Figure 3).
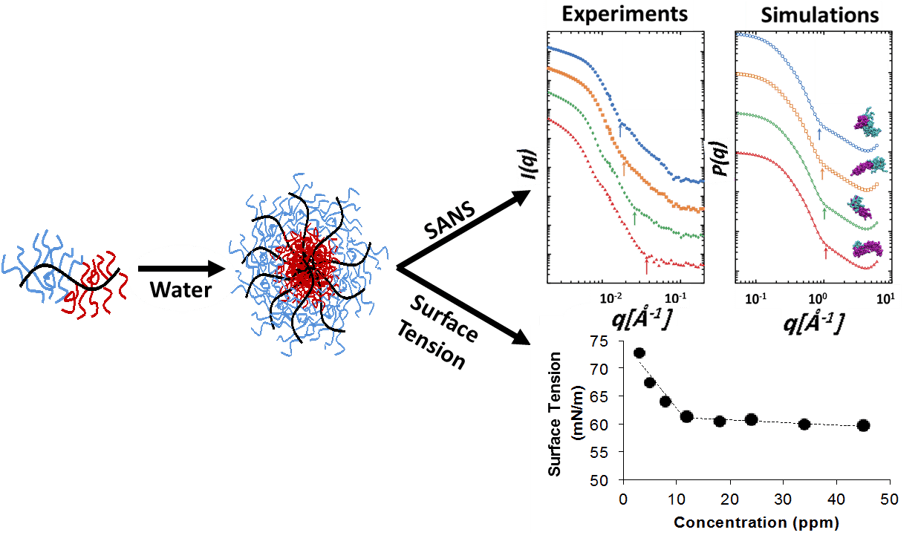
Figure 3. Self-assembly of bottlebrush block copolymers in water. Reproduced from Alaboalirat, M.; Qi, L.; Arrington, K. J.; Qian, S.; Keum, J. K.; Mei, H.; Littrell, K. C.; Sumpter, B. G.; Carrillo, J-M, Y.; Verduzco, R. Matson, J. B. “Amphiphilic Bottlebrush Block Copolymers: Analysis of Aqueous Self-Assembly by Small Angle Neutron Scattering and Surface Tension Measurements” Macromolecules, 2019, 52, 465-476.
New Renewable and Degradable Polymers:
New degradable materials made from renewable feedstocks are desperately needed. We aim to apply modern polymer chemistry to old polymers, mostly polysaccharides, to make new copolymers and polymer blends that have useful properties. In this project, we work closely with the group of Kevin Edgar (VT Sustainable Biomaterials). For example, we prepared ABA triblock copolymers of the structure CTA-b-PB-b-CTA (CTA = cellulose triacetate; PB = poly(cis-1,4-butadiene) using a combination of CTA end-group functionalization and ROMP. We then found that these block copolymers were effective compatibilizers for blends of CTA and PB, increasing their toughness over 60-fold compared with uncompatibilized blends (Figure 4).
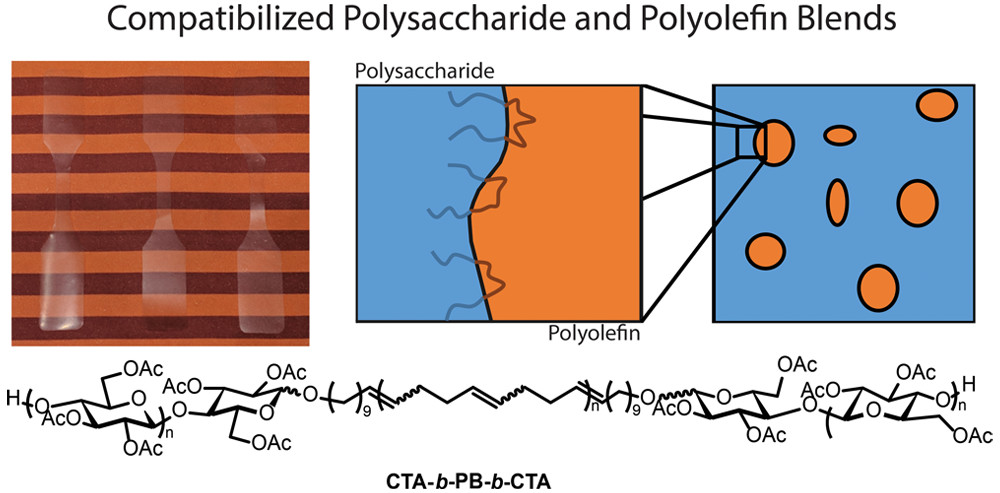
Figure 4. Images of films and cartoon of compatibilized CTA/PB blends using the ABA triblock copolymer compatibilizer shown. Reproduced from Arrington, K. J.; Haag, J. V.; French, E.; Murayama, M.; Edgar, K. J.; Matson, J. B. “Toughening Cellulose: Compatibilizing Polybutadiene and Cellulose Triacetate Blends” ACS Macro Lett., 2019, 8, 447-453.
Self-Assembling Peptide-Based Materials:
Amphiphilic peptides are capable of self-assembling in aqueous solution into unique and beautiful nanostructures. We recently discovered a set of aromatic peptide amphiphiles based on simple tetrapeptides that form regular nanohelices in water (Figure 5). These nanocoils are cylindrical nanofibers that take on a left-handed helical conformation, as revealed by transmission electron microscopy in collaboration with the group of Ronit Bitton (Ben Gurion University in Israel). They are capable of releasing H2S in response to a cysteine trigger, and we have shown that they can rescue heart cells from the toxic effects of common cancer chemotherapeutics. Through coarse-grained molecular dynamics simulations with the group of Sanket Deshmukh (VT Chemical Engineering), we discovered that these tetrapeptide nanocoils have closely spaced glutamic acid residues, which we have found enables efficient ion conduction in water. We are also investigating these nanocoils as catalytic nanostructures and as materials capable of templating the formation of nanoparticles.

Figure 5. Chemical structure (A), cryogenic transmission electron micrograph (B) and atomic force micrograph (C) of self-assembling amphiphilic peptides. Reproduced from Wang, Y.; Kaur, K.; Scannelli, S. J.; Bitton, R.; Matson, J. B. “Self-Assembled Nanostructures Regulate H2S Release from Constitutionally Isomeric Peptides” J. Am. Chem. Soc. 2018, 140, 14945-14951.